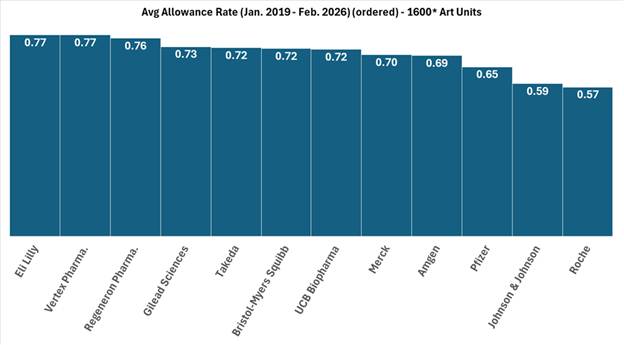
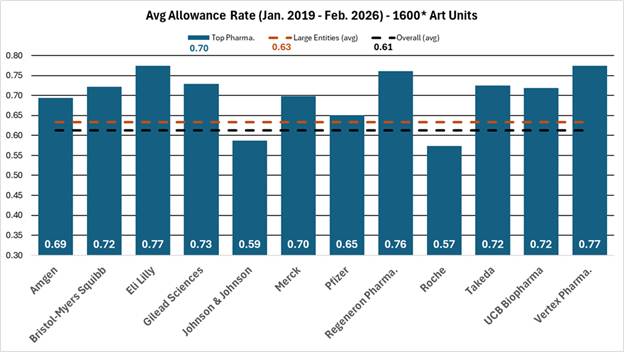
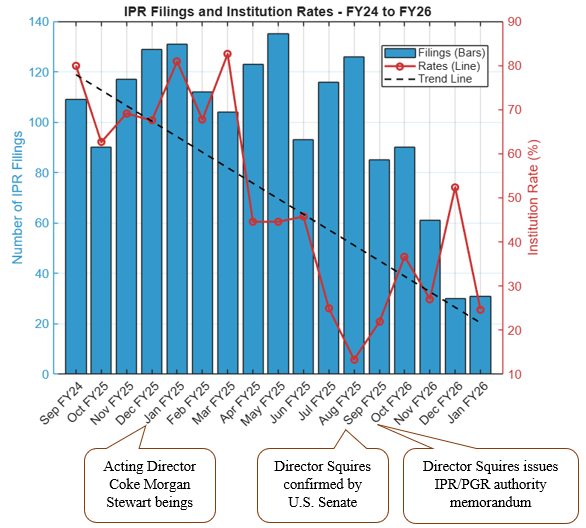
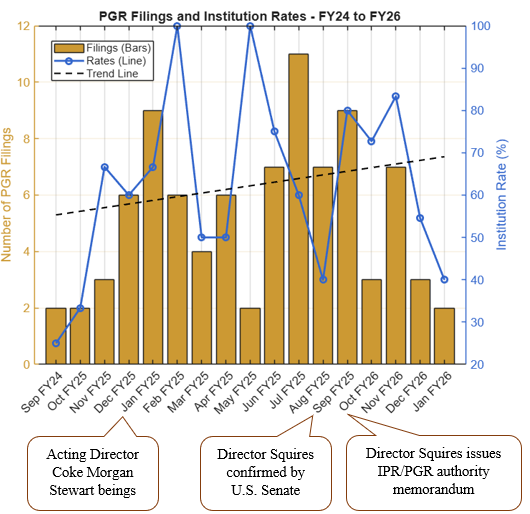
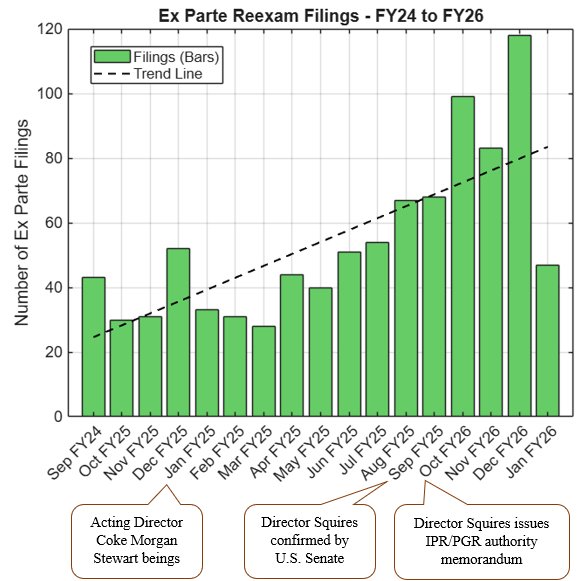
The Supreme Court heard oral argument on April 29, 2026, in Hikma Pharmaceuticals USA Inc. v. Amarin Pharma, Inc. (No. 24-889), the closely watched skinny-label case for which the Court granted certiorari earlier this year. As discussed in our prior post, Supreme Court to Address ‘Skinny Label’ Patent Dispute,the case has significant implications for generic manufacturers navigating carve-out labeling strategies under Hatch-Waxman.
The case marks the Supreme Court’s first patent matter since Amgen Inc. v. Sanofi and could substantially affect how courts evaluate induced infringement claims involving skinny labels.
Brief Recap of the Dispute
Amarin is the maker of Vascepa® (icosapent ethyl), a drug originally approved to treat severe hypertriglyceridemia (very high triglycerides). Amarin later invested approximately $300 million in the landmark REDUCE-IT clinical trial, which demonstrated that Vascepa® also reduces the risk of cardiovascular events — a blockbuster new indication that generated over a billion dollars in sales. Amarin obtained later-expiring method-of-use patents covering this cardiovascular indication.
The dispute arises from Hikma’s generic version of Vascepa®. Hikma launched its product with an FDA-approved skinny label that carved out Amarin’s patented cardiovascular indication while retaining approval for severe hypertriglyceridemia.
Amarin nevertheless alleged that Hikma induced infringement of its patents through certain public statements and marketing materials that allegedly encouraged physicians to prescribe the product for the patented cardiovascular use. In particular, Amarin pointed to Hikma press releases referring to the product as a “generic version of Vascepa” and referencing Vascepa’s overall sales figures, including sales associated with the patented cardiovascular indication.
Although the district court dismissed the claims, the Federal Circuit revived the case in 2024, concluding that Amarin plausibly alleged induced infringement when considering Hikma’s labeling and related statements together. Hikma petitioned the Supreme Court, arguing that the Federal Circuit’s approach threatens the viability of the Section viii carve-out framework and creates uncertainty for generic manufacturers relying on skinny labels.
Oral Argument Focused on Limits of Induced Infringement
The April 29, 2026, oral arguments brought into focus how far inducement liability can extend when a generic drug is launched under a Section viii “skinny label” carve-out. The Court heard from Hikma, Amarin, and the United States, which appeared as amicus curiae supporting Hikma (the petitioner) and was allotted separate argument time given the case’s implications for the federal Hatch-Waxman framework.
During the hearing, the Justices appeared focused on identifying an appropriate standard that balances inducement liability with Congress’ intent to permit carved-out labels for unpatented uses under the Hatch-Waxman framework. Several members of the Court questioned whether allowing liability based on combination of labeling and surrounding communications risk undermining the statutory safe space created by the skinny-label pathway, particularly given the significant cost savings associated with generic drugs. The Court also appeared concerned with an appropriate evidentiary threshold for inducement liability, including whether generalized marketing references and routine commercial communications are sufficient to establish liability or whether more specific proof of encouragement is required.
Hikma contended that inducement requires clear, affirmative encouragement of infringing use, not inferences drawn from FDA-compliant labeling or routine marketing. It emphasized that the Section viii framework is designed to preserve lawful non-patented uses and cautioned that treating ordinary FDA-compliant marketing language as evidence of intent to induce infringement would effectively undermine the statutory-carve out. Hikma painted a stark picture of the consequences, arguing that under the Federal Circuit’s decision, a generic can get FDA approval of a skinny product label that is required by statute to mirror the brand’s label, but as soon as the product is commercialized and described as a generic version, even with disclaimers, it creates infringement liability. Hikma pointed to a prior case involving GSK and Teva where “Teva got hit for hundreds of millions of dollars that dwarfed its actual profits.”
The United States largely agreed with Hikma, stressing that a skinny label is federally mandated and therefore cannot, by itself, support an inference of inducement. Deputy Solicitor General Malcolm L. Stewart argued that the Federal Circuit erred by giving weight to FDA-required labeling or regulatory compliance, cautioning that this approach risks converting lawful conduct into evidence of liability. He further warned that an inference-based standard could deter generic entry by exposing manufacturers to inducement claims despite staying within FDA approval, thereby undermining the Hatch-Waxman scheme threatening the availability of lower-cost generic drugs. Stewart emphasized a foundational principle: “It’s a bedrock principle of inducement law that active inducement to infringe is required. The question is not whether the defendant has adequately warned people away from infringement.” Stewart also explained — in response to questioning from Justice Jackson on why describing a product as “the generic version of Vascepa” is innocuous: “Under the abbreviated new drug application process, the generic comes on the market not by conducting independent studies of safety and efficacy but by showing that it is equivalent to a drug that’s already been found safe and efficacious. And so, when Hikma says we’re the generic version of Vascepa, they just mean Vascepa is what’s called the reference listed drug.” Responding to questions about broader market implications, Stewart posited that “we certainly think that if the Federal Circuit’s analysis were affirmed, that it would create a substantial disincentive to entering the generic market.”
Amarin countered that inducement can be shown through circumstantial evidence, especially at the pleading stage. It argued that Hikma’s references to Vascepa®, along with marketing language suggesting interchangeability and highlighting full-market potential, plausibly encouraged physicians to prescribe the generic for the still-patented indication. In Amarin’s view, FDA compliance does not shield a manufacturer where broader promotional conduct allegedly encourages infringing use. Amarin argued that: “If a branded drug like Amarin cannot get any kind of protection for a newly discovered use of its existing drug, it would just be economically irrational to make that kind of investment that discovers a lifesaving cure. And I think that’s what we are really fighting for in this case, is the opportunity to say: We spent five years, we spent $300 million, and we discovered something that is literally saving people’s lives. Hikma wants to come and, having spent no money basically at all, try to capture all of those gains.” In response to Congressman Waxman’s amicus brief stating that “the Federal Circuit’s decision threatens to decimate the compromise at the heart of the Hatch-Waxman Act” and that “generics have saved 3.4 trillion over the past 10 years, but the Federal Circuit’s decision here leaves generic drug companies in the dark about what might expose them to liability.” Hikma responded by emphasizing that the other side of the Hatch-Waxman compromise was “about protecting the need to encourage branded drugs to take existing products and invest massive resources to discover how those drugs can be used for new cures.”
Implications for Generic Competition Framework and Beyond
Although the Hikma v. Amarin arises under Hatch-Waxman, the Court’s decision could influence how courts evaluate induced infringement claims involving carved-out indications across the pharmaceutical sector. A broader inducement standard could increase litigation exposure for manufacturers that rely on skinny labels to enter markets with partially patented products. This risk may be particularly serious where allegations rely on a combination of FDA-approved labeling and non-label communications such as press releases or product descriptions. Conversely, a narrower standard could reinforce the predictability of the Section viii carve-out mechanism and preserve its role as a key pathway for earlier generic entry.
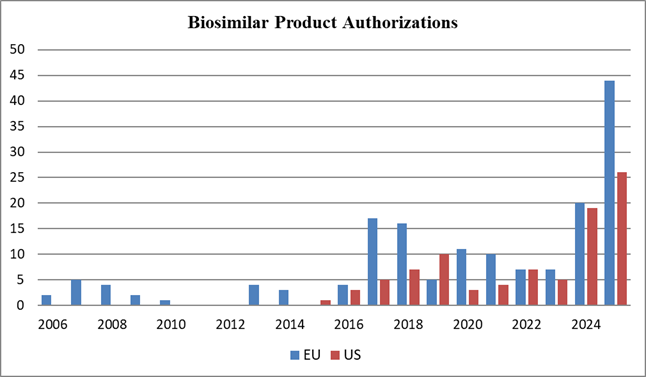
Similar issues may also emerge in the biologics context under the Biologics Price Competition and Innovation Act of 2009 (BPCIA), which not only provide an abbreviated approval pathway for biosimilars but also creates a defined process for resolving patent disputes between biosimilar sponsors and reference product sponsors. In particular, the breadth of the inducement standard may influence how courts assess biosimilar launch communications and indication-specific labeling strategies in infringement disputes.
Looking Ahead
The most contentious factual question is whether repeatedly calling a product “generic Vascepa” at a time when Vascepa® was famous for its cardiovascular benefits — plausibly constitutes active encouragement of the patented use, or whether it is merely an accurate product description (reference listed drug identification) with an obvious non-infringing explanation.
Procedural issues also loom if the Supreme Court were to remand, e.g., whether to vacate the Federal Circuit’s opinion and remand to the Federal Circuit, which would remand it to the district court to permit Amarin to amend its complaint, or because the dismissal was with prejudice and Amarin conceded to that judgment, that any remand would leave only a Rule 60 motion, essentially declaring the case over save for exceptional circumstances.
A decision is expected later this Term, and the Court’s ruling could become a defining precedent for induced infringement in the pharmaceutical context. Regardless of outcome, Hikma v. Amarin is likely to be one of the most consequential pharmaceutical patent decisions in recent years, particularly for manufacturers navigating the intersection of FDA labeling pathways and patent enforcement strategies. We will continue to monitor further developments relating to this case and provide an update when the Court issues its decision.
Disclaimer: The information contained in this posting does not, and is not intended to, constitute legal advice or express any opinion to be relied upon legally, for investment purposes or otherwise. If you would like to obtain legal advice relating to the subject matter addressed in this posting, please consult with us or your attorney. The information in this post is also based upon publicly available information, presents opinions, and does not represent in any way whatsoever the opinions or official positions of the entities or individuals referenced herein.